
One major problem with Bohr's model was that it could not explain the properties of atoms with more than one electron, and by the early 1920s, the search for a new atomic model had begun. 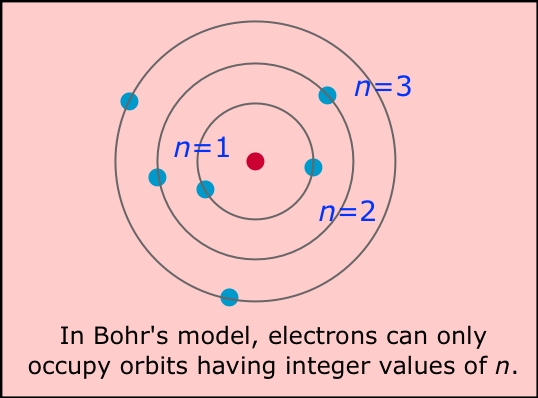
Bohr's atomic model was very stable because the electron could not lose any more energy than it had in the smallest orbit. In 1913 Bohr proposed his quantized shell model of the atom to explain how electrons can have stable orbits around the nucleus. Published in 1913, Bohrs model improved the. Atomic Theory Model based on the Kojic Acid Serum Diy Atomic Theory Timeline Henry Moseley (1887-1915. The Bohr model of atomic structure was developed by Danish physicist and Nobel laureate Niels Bohr (18851962). energies for the Bohr atom : This n is known as the principal quantum number. Bohr was able to calculate the energy of the electron in a hydrogen atom by measuring the wavelengths of the light emitted in its line-emission spectrum. Quantum Theory of the Hydrogen Atom 6.1 Schrödingers Equation for the Hydrogen Atom Todays lecture will be all math. The 6 protons and 6 neutrons are housed in the carbon atom’s nucleus while the 6 electrons are filled in the two shells, i.e., K and L shells, as 2 and 4, respectively. The energy of each photon corresponds to a particular frequency of radiation given by Planck's equation, E = hλ. Therefore, the final Bohr model of carbon appears as follows: Hence, the final Bohr model of the Carbon atom consists of 6 protons, 6 neutrons, and 6 electrons. The energy of the photon is equal to the energy difference between the two orbits. When an electron falls from the excited state to the ground state, a photon is emitted with a specific energy. The electron gains or loses energy by moving to a new orbit, either further from or closer to the nucleus. While in an orbit, the electron has a fixed amount of energy. Bohr was the first to discover that electrons travel in separate orbits around the nucleus and that the number of electrons in the outer orbit determines the properties of an element. 
Likewise, the electrons of an atom can occupy one orbit or another, but cannot exist in between orbits. The Bohr model shows the atom as a small, positively charged nucleus surrounded by orbiting electrons. As a person climbs up a ladder, they step on one rung or another, but not in between rungs, because a person cannot stand on air. These orbits can be thought of as the rungs of a ladder.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
